Boehringer’s stake in the firm equates to around 16.4%
News
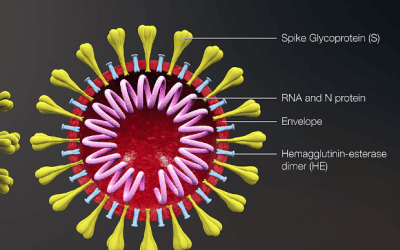
New coronavirus saliva test to be trialled in Southampton
Over 14,000 GP staff, other essential key workers and university staff and their households will participate in the first phase of the trial
MHRA extends access to Santhera’s DMD drug via the EAMS
The scientific opinion has been extended for one year while the drug is under regulatory review
Gilead to start testing an inhaled form of remdesivir
The firm is targeting an earlier stage in the treatment pathway for COVID-19
Last chance to register for the Patient Partnership Index 2020
Registration will close June 23 but entry forms can be submitted until July 3
GSK/Clover’s COVID-19 vaccine moves into human trials
The Phase I study is assessing use of a GSK pandemic adjuvant in combination with COVID-19 vaccine candidate SCB-2019
Novartis pulls plug on hydroxychloroquine trial
Study assessing its potential for COVID-19 discontinued due to feasibility of recruitment
ABPI consults on ‘new-look’ Code of Practice
A new version of the Code is expected to come into force in July 2021
EMA to consider expanding use of Merck/Pfizer’s Bavencio
The regulator will assess its safety and efficacy as a treatment for bladder cancer
Novo Nordisk’s AM833 shows early success in obesity trials
The firm is ‘encouraged’ by the performance of the drug, both as monotherapy and in combination with semaglutide
EU filing for CPP’s familial adenomatous polyposis drug
The drug prevented more than 90% of precancerous sporadic adenomas in trials
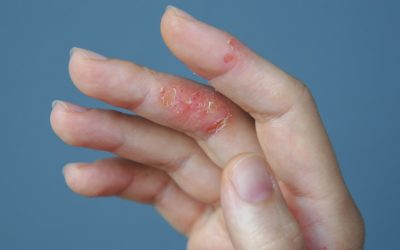
AbbVie’s Rinvoq shows potential for atopic dermatitis
The selective and reversible JAK inhibitor hit all goals in the Phase III Measure Up study
Waldenström’s macroglobulinemia drug Brukinsa accepted for EU review
WM is a rare lymphoma representing around 1% of all non-Hodgkin lymphomas
High-street pharmacist check-ups overshoot 300,000
Local pharmacies now gearing up to provide an increasing number of consultations as people encouraged to seek healthcare advice if needed
Roche’s Tecentriq shows promise in early triple-negative breast cancer
Less patients who received the Tecentriq combination before surgery had evidence of tumour tissue detectable at the time of surgery