The European Commission has approved Novartis’ Rydapt to treat a certain form of acute myeloid leukaemia (AML) as well as three other rare diseases.
News
NICE backs use of Opdivo for lung cancer
Bristol-Myers Squibb’s Opdivo is being recommended by the National Institute for Health and Care Excellence to treat a certain form of lung cancer on the NHS in England, via the Cancer Drugs Fund.
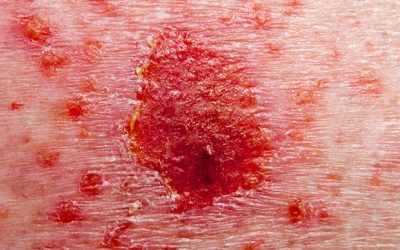
Janssen’s guselkumab shows long-term benefit in psoriasis
Janssen has unveiled new data showing consistent rates of skin clearance with guselkumab treatment among patients with moderate to severe plaque psoriasis.
Teva sells off women’s health assets for $1.38bn
Teva has sold the remaining assets in its speciality global women’s health portfolio for $1.38 billion.
GSK, Innoviva announce US nod for Trelegy Ellipta
GlaxoSmithKline and Innoviva have bagged a green light in the US for the novel triple therapy inhaler Trelegy Ellipta, opening the door to a new treatment option for patients with Chronic Obstructive Pulmonary Disorder (COPD).
AbbVie/Roche’s venetoclax hits goals in CLL trial
A combination of AbbVie and Roche’s venetoclax and rituximab has shown a progression-free survival benefit in patients with chronic lymphocytic leukaemia (CLL).
CHMP backs 13 treatments for EU approval
Thirteen new therapies – including two biosimilars – have been backed for approval in Europe.
Bavarian Nordic hit by cancer immunotherapy failure
Bavarian Nordic saw its shares plummet as much as 50 percent in the wake of news that the firm’s cancer immunotherapy ProstVac failed to prolong survival in men with a particular type of prostate cancer.
Bayer’s rare cancer drug Aliqopa bags speedy approval
US regulators have issued an accelerated approval for Bayer’s relapsed follicular lymphoma therapy Aliqopa.
NICE rejects first-line use of Roche’s Gazyvaro
The National Institute for Health and Care Excellence has turned down first-line use of Roche’s Gazyvaro in patients with untreated advanced follicular lymphoma on the NHS in England and Wales.
Marketer & Communications Team of the Year – venue announced!
This year’s gala awards and dinner will take place at the London Marriott Hotel Grosvenor Square in Mayfair on Thursday 9 November.
Five firms named in breaches of ABPI code
Astellas UK, Astellas Europe, Pfizer, Novartis Pharmaceuticals and Tor Generics named in advertisements
Most HCPs not aware of Accelerated Access Review – report
Many are also unaware of other initiatives to improve adoption of innovation
FDA approves first cancer biosimilar
Amgen’s Mvasi is a biosimilar of Genentech’s Avastin
Number of written complaints about NHS increases 4.9 per cent
208,400 written complaints received during 2016/17