At 48 weeks, 98% of patients treated with the drug were relapse free compared to 63% of patients receiving placebo.
News
New cancer research could help boost immunotherapy treatments
Scientists have found a way to pull down the protective wall that surrounds tumours, potentially re-exposing them to the killing power of the immune system.
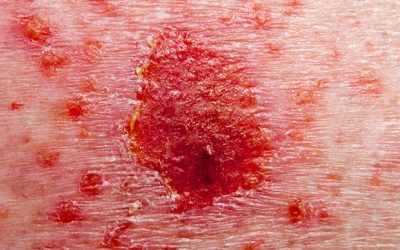
Lilly’s atopic dermatitis drug hits endpoints
Both the 4mg and 2mg dosages met the trials’ primary endpoint in BREEZE-AD1 and BREEZE-AD2.
NICE to work with GW Pharma after Epidiolex rejection
The committee also cited concerns about the economic models provided by the company.
Positive multiple myeloma results for GSK
Belantamab mafodotin is on track for regulatory submission by the end of 2019.
FDA Fast Track granted for AZ’ Forxiga
The indication, chronic kidney disease, is a serious, progressive condition defined by decreased kidney function.
EMA accepts license extension submission for Invokana and Vokanamet
The decision is based on results from the landmark Phase III CREDENCE study
Multiple antibiotic courses linked to pediatric treatment failure
Data showed that those who had been given at least two previous courses were 32% more likely to fail to respond to a subsequent course.
NICE recommends antiseptics over antibiotics for impetigo
Antiseptic such as hydrogen peroxide 1% cream is ‘just as effective’ as a topical antibiotic.
HSE reimburses Braftovi combo
Eligible patients in the Republic of Ireland can potentially benefit from the combination with immediate effect.
One in seven five-year-olds don’t have MMR jabs
The lack of immunisation leaves children at high risk of measles at a time when outbreaks of the disease are occurring across the country.
Priority Review awarded to Xtandi for prostate cancer
Back in February, the companies revealed results from the Phase III ARCHES trial in men with mHSPC.
Fiasp approved for children in Europe
Novo Nordisk submitted label updates in March this year.
NICE chief executive to step down
Sir Andrew Dillon has led the organisation for 20 years.
Skyrizi bags NICE fast-track
Eligible patients in England and Wales will have access to the drug almost two months earlier.