The “state of the art” facility will be the new base for 500 of the 1,200 employees in the UK.
News
GSK recalls prescription Zantac
The FDA claims to have found “unacceptable” levels of carcinogen in the medicine.
Cuts in alcohol duty linked to 2000 deaths
The knock-on effects of these cuts has also seen an increase of over 61,000 hospital admissions since 2012 at an estimated cost of £317m to the NHS.
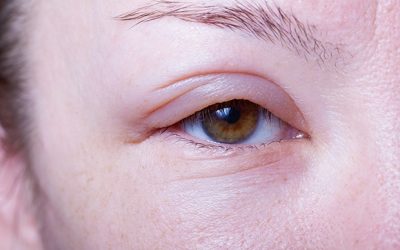
1/3 of people with sight loss suffer mental health problems
84% of Brits fear losing their sight more than any other sense.
Bayer, Sensyne to form new AI ‘LifeHub’
Earlier this week Sensyne announced a formal research agreement with the MHRA.
Elmiron bags draft NICE approval
The recommendation comes after Consilient Health agreed a Patient Access Scheme with NICE.
Avacta, ADC Theraputics sign collaboration and option agreement
The partnership will focus on developing potent Affimer-drug conjugates.
MS Society launches public appeal for £100m funding
The Stop MS Appeal aims to raise the funds over ten years to find treatments for everyone living with the progressive, immune-mediated disorder.
Ligelizumab trumps Xolair in spontaneous urticarial trial
The drug achieved complete control of hives in 51% and 42% of patients, compared with 26% of patients treated with Xolair.
Tickets available for the 2019 Communications Team of the Year gala dinner & awards ceremony
This year’s awards ceremony will be taking place at the Chelsea Harbour Hotel on the 14th November.
NHS to roll out ‘whistle-blower’ scheme
The scheme will offer practical support to any doctor, nurse, or other worker across the country who needs additional support to build their career after raising concerns at work.
GSK, Lyell join forces for cancer cell therapies
The partnership will develop new technologies to improve cell therapies for cancer patients.
Sprifermin shows promise in knee osteoarthritis
There are no approved drugs at current for preventing or slowing disease progression.
Theragnostics, GE Healthcare announce prostate cancer diagnostic partnership
Theragnostics will lead the development of the tracer, GalliProst, while GE Healthcare will lead all pre-approval commercial preparations.
Benepali bags positive psoriasis results
Samsung Bioepis has announced real-world data from the drug.