“A positive trend in overall survival” had been seen in a trial, with an interim analysis demonstrating a 29% reduction in the risk of death for Nubeqa.
News
LEO issues Picato recall
Last September, the EMA’s Pharmacovigilance Risk Assessment Committee announced a review of data on skin cancer in patients using the drug.
GSK hit with EU Court ruling over ‘pay-for-delay’ drug case
The ruling harks back to the CMA’s 2016 case that found GlaxoSmithKline had agreed to make payments totalling over £50 million to other generic suppliers of paroxetine.
Peanut allergy vaccine shows promise
There are currently no approved immunotherapies for the treatment of peanut allergies.
New drug candidate created by AI in ‘world first’
The drug, DSP-1181, was created through joint research by both the companies and Centaur Chemist’s AI platform.
J&J launches ‘multi-pronged’ coronavirus response
Using its AdVac and PER.C6 technologies, the company wants to rapidly upscale production of the optimal vaccine candidate.
NHS to tackle climate “health emergency” with new campaign
The campaign, dubbed “For a Greener NHS”, will address both the causes of climate change and air pollution.
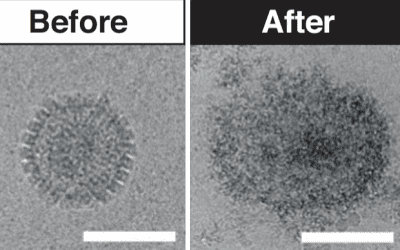
New treatment ‘made from sugar’ could help fight viral outbreaks
The team has “successfully engineered a new molecule, which is a modified sugar that shows broad-spectrum antiviral properties.”
EMA validates Kite’s CAR-T filing
In December last year the company also filed the CAR-T therapy with the FDA.
Industry leading companies are entering their best and brightest in CROY 2020
The 2020 competition has already seen a large number of top industry companies being represented as we approach the final month of entries.
European Commission expands use for Janssen’s Erleada
The extension is based on data from the Phase III TITAN study, which assessed the addition of the drug to androgen deprivation therapy in a broad range of patients with metastatic hormone-sensitive prostate cancer.
Oncimmune test more effective than CT Surveillance in lung cancer
Lung cancer is one of the most prevalent forms of cancer and kills more women than breast and ovarian cancers combined.
NHS competition to inject £140m in to ‘game changing’ AI
The competition entries are open from Tuesday 28 January for the next five weeks, and companies can bid for a share of £140 million to launch their innovation across the health service.
Cancer patients facing delays in getting ‘innovative’ drugs
A study found that the most innovative cancer drugs took 3.2 years longer to go from the filing of the patent through to NHS.
‘Rapid’ NHS response teams to help older people at home
Backed by £14 million of investment, local health service and council teams will begin the roll out of the “Urgent Community Response” teams from April.