Compared to placebo, the drug didn’t achieve significant reduction in motor and phonic tics.
News
Half of GPs skip out ‘vital’ diabetes kidney test
The test, which is used to detect kidney damage that can lead to fatal complications, should be conducted every year.
Roche’s Tecentriq bags Priority Review in US
In a trial, the drug improved overall survival by 7.1 months compared with chemotherapy.
US breakthrough designation granted to Padcev in bladder cancer
The designation was granted based on results from the dose-escalation cohort and expansion cohort A of the EV-103 Phase Ib/II trial.
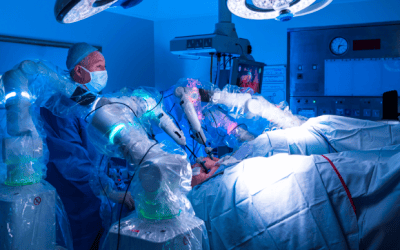
‘Transformative’ surgical robot trialled on NHS
The step marks a “pivotal milestone” for Versius.
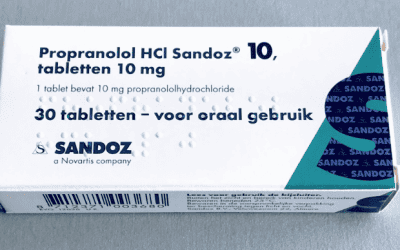
New report highlights ‘toxic’ risk of propranolol
There was a 34% increase in overdose deaths caused by propranolol between 2012 and 2017, and by 2016 nearly 4.7 million prescriptions were issued to patients annually.
Opdivo, cabiralizumab combo falls short in pancreatic cancer
Bristol-Meyers Squibb said that it will continue to support the evaluation of cabiralizumab in select, ongoing investigator-sponsored trials.
Vyndaqel first treatment approved in Europe for ATTR-CM
The decision was based on results from the Phase III ATTR-ACT study.
Sanofi to repurpose SARS vaccine candidate for coronavirus
The company joins J&J, Gilead and GSK in the ranks searching for a vaccine for the global health epidemic.
First patient undergoes Luxturna gene therapy on NHS
Jake Ternet, patient at Moorfields Eye Hospital was the first in the UK to receive the treatment.
MS Society launches ‘first-of-its-kind’ fatigue course
The Society hopes that the new online function will relieve pressure on healthcare professionals, and could result in significant cost savings for the NHS as demand for the face-to-face programme lessens.
New technique developed to ‘listen in’ on cancer cells
Understanding how cells communicate could reveal how tumours are able to evade the immune system and become resistant to treatments.
Scientists create ‘most detailed’ breast cancer map ever
To develop the maps, the team looked within 483 different tumour samples for the presence of 37 key proteins.
Beovu bags EU approval in wet AMD
The drug is the first EC-approved anti-VEGF treatment to demonstrate superior resolution of retinal fluid, compared to Bayer’s Eylea.
Arctoris, Molecule join forces on drug discovery
The news comes as drug discovery rates are declining, the price of bringing a drug to market is increasing, and return on investment (ROI) on R&D is expected to hit 0% in the next few years.