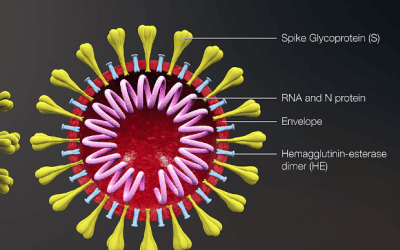
Sense Biodetection has also pitched in, announcing an accelerated programme to launch the world’s first instrument-free, point-of-care molecular diagnostic test for the novel virus.
News
FDA approves Imfinzi for extensive-stage SCLC
The approval was based on the Phase III CASPIAN trial, in which the risk of death was reduced by 27%.
Nubeqa authorised in Europe for certain prostate cancer patients
The European authorisation is based on the Phase III ARAMIS trial.
Ensifentrine improves lung function in COPD patients
However, the company has decided to postpone the initiation of Part B due to concerns regarding the COVID-19 pandemic.
Brilinta found to reduce bleeding vs dual therapy
Acute coronary syndrome is a type of cardiovascular disease that occurs when a blood clot forms.
COVID-19: NICE updates guideline as Sanofi announces vaccine development
Vertex has also revealed its plans to keep clinical trials going during the unstable time.
CHMP backs eight new meds in virtual March meeting
The meeting was held virtually to help limit the spread of COVID-19.
Forxiga successful in DAPA-HF heart failure trial
The new data from the trial “further reinforce Forxiga’s clinical effects beyond diabetes.”
AZ recruits ImaginAb in CD8 ImmunoPET deal
CD8 ImmunoPET minibody works by binding CD8 receptor on human T cells and is used for non-invasive PET imaging of CD8 T cells in patients.
COVID-19: UK commits £210m as pharmacist warns Brits not to stockpile Ventolin inhalers
Thermo Fisher has also announced that it has received the CE mark in the European Union for its diagnostic test.
CHMP gives Sanofi positive opinion for Sarclisa
Despite available treatments, multiple myeloma remains an incurable malignancy, and is associated with significant patient burden.
BMJ journal deems opioid dependency implant more favourable than tablets
While the opioid epidemic in the USA is well documented, Europe also has an estimated 1.3 million high-risk opioid users.
Takeda, Evox link up to the tune of $882m
Evox will offer its preclinical programme in Niemann-Pick disease type C for development, and a second new programme directed at another undisclosed rare disease.
EMA accepts Arvelle’s cenobamate MAA
There are about six million people in Europe with epilepsy, and approximately 40% of adults with focal-onset seizures continue to experience seizures.
COVID-19: ‘Army’ of volunteers join NHS as BMS postpones new study starts
The FDA has also granted Gilead’s wishes to withdraw orphan drug designation for remdesivir under current circumstances.