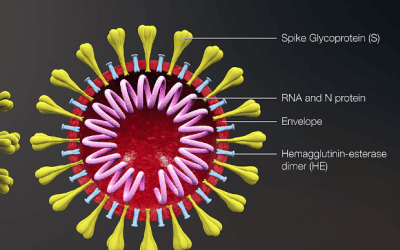
If approved, lumasiran would be the first approved therapeutic treatment option for primary hyperoxaluria type 1
News
Vertex Foundation donates funds to support COVID-19 efforts
The move falls under a $5-million global programme to support the COVID-19 pandemic response around the world
Cancer Research UK cuts fundings grants amid pandemic uncertainty
The cuts equate to £44 million, driven by a loss in fundraising income
GSK links with Vir Biotechnology to find coronavirus solutions
GSK has bought a $250 million stake in the firm
Firms form alliance to work on COVID-19 therapy
The alliance will immediately start work on the development of an unbranded anti-SARS-CoV-2 polyclonal hyperimmune immunoglobulin medicine
Imophoron develops COVID-19 vaccine candidates
Preclinical testing expected “within weeks”
Menlo hit with serlopitant setback
Shares in the firm dropped nearly 50% in pre-market trading
UK PM Boris Johnson in intensive care
The prime minister remains conscious and not on a ventilator at this time, official say
UK medical chiefs urge recruitment in COVID-19 trials
A letter has been sent to all NHS trusts urging efforts to enrol COVID-19 patients in trials
NICE publishes four more rapid guidelines in response to pandemic
The guidelines cover active management of patients with a number of conditions as well as COVID-19 in the community setting
Izana intiates study assessing namilumab for COVID-19
It is hoped that anti-GM-CSF therapies such as namilumab might play an important role in preventing or reducing deterioration in COVID-19 patients
UK’s prime minister Boris Johnson admitted to hospital
The move is a ‘precautionary measure’, Downing Street officials have stressed
EU nod for Novo’s diabetes drug Rybelsus
Launches in the first EU countries are expected to take place in the second half of 2020
GSK’s Nucala shows promise in nasal polyps
Nucala is the first anti-IL5 biologic to report positive Phase III results in patients with the condition says GSK
Pfizer wins EU approval for MabThera biosimilar
The drug will be available to treat certain cancers and autoimmune conditions