Drug will be launched in the UK first
News
GW Pharma begins recruitment for US-based Sativex trial
Trial will evaluate Sativex (nabiximols) for multiple sclerosis spasticity
Bristol Myers Squibb’s psoriasis med hits the mark in phase III
Deucravacitinib demonstrated superiority to placebo and Otezla in phase III
MHRA starts rolling review of AZ’s coronavirus vaccine
British drugmaker confirmed review on Sunday
Oxford BioDynamics enters strategic partnership with Boca Biolistics
Partnership aims to advance disease severity testing programme for COVID-19
EMA begins evaluation of filgotinib for ulcerative colitis
Gilead and Galapagos’ application for oral JAK1 inhibitor validated by agency
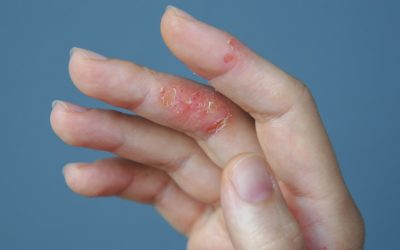
Lilly, Incyte tout new baricitinib data in eczema
Data presented at virtual European Academy of Dermatology and Venereology congress
Novo Nordisk’s Saxenda bags NICE recommendation
Obesity management treatment recommended following confidential discount
New study assessing antibiotics for COVID-19 sepsis
It is hoped that findings from the NIHR-funded trial will help better inform treatment decisions
Sanofi to buy Kiadis
The move gives the French drug giant access to Kiadis’ natural killer cell technology platform
MHRA allows HAE patients early access to berotralstat
The oral, once-daily therapy is designed to prevent attacks and reduce burden of therapy
New EU approval for GSK’s Zejula
Zejula is the first PARP inhibitor to be approved as monotherapy in the EU for patients with platinum-responsive advanced ovarian cancer, regardless of biomarker status
Blood test more predictive of ovarian cancer than ‘previously thought’
Findings come from new research funded by Cancer Research UK and NIHR
AZ’s Fasenra hits the mark in phase IIIb asthma trial
Drug significantly eliminated oral corticosteroid (OCS) in OCS-dependent asthma patients
UK biotech consortium identifies COVID-19 antibody combinations
The UK BIA Antibody Taskforce is set to take combinations into further development as a cocktail