Positive recommendation follows initial rejection in September
News
FDA approves new dosing option for AZ’s Imfinzi
Less-frequent, fixed-dose use approved for two cancer indications
Sanofi’s meningococcal vaccine wins EU approval
MenQuadfi approved for vaccination in individuals 12 months of age or older
Annual pharma R&D spending in UK climbed 6.9% in 2019
R&D spending in the sector last year represents an eight-year high
Lilly to collaborate with Precision BioSciences on genome editing research
Lead programme will focus on developing therapies for Duchenne muscular dystrophy
Spending boost ‘welcome’ but more funding needed, says NHS Confed chief exec
Health leaders respond to reported £3bn funding boost
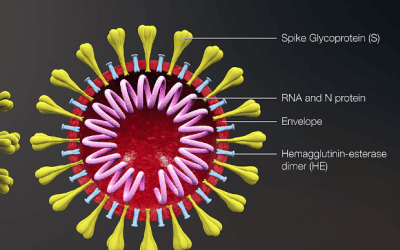
AZ/Oxford University’s COVID-19 vaccine shows average efficacy of 70%
Effectiveness ranges from 62% to 90% depending on dosing regimen
Amazon launches online pharmacy in the US
Service also offers price comparison on generic and branded drugs
Alnylam’s RNAi therapeutic Oxlumo gets EU green light
Treatment approved to treat primary hyperoxaluria type 1
Sosei Heptares’ COVID-19 programme advances promising molecules
R&D programme is focused on developing small molecules against SARS-CoV-2 virus
Novartis secures rights to Mesoblast’s remestemcel-L for ARDS
Drug is currently being evaluated in a phase III COVID-19-related ARDS study
Novel antibiotic Recarbrio has launched in the UK
Combination treatment scored EU approval earlier this year
Multiple sclerosis trial focusing on people who can’t walk begins recruitment
Trial will evaluate whether Mavenclad can slow the rate of upper limb disability progression
NICE recommends Lilly’s migraine med Emgality
CGRP antagonist approved to prevent migraine in adults with episodic and chronic migraine
AZ’s COVID-19 vaccine produces ‘encouraging’ immune response in older adults
New findings from phase 2 study published in The Lancet