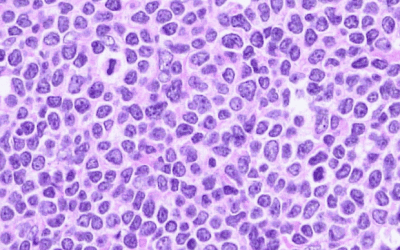
Therapy approved for the treatment of paediatric patients with anaplastic large cell lymphoma
News
UK biotech Ixaka scores additional funding for cell and gene therapy research
Lead asset REX-001 is in Phase III development for chronic limb-threatening ischaemia
AZ, Daiichi Sankyo’s Enhertu wins US approval for HER2-positive gastric cancer
Approval is based on results from the Phase III DESTINY-Gastric01 trial
Boehringer Ingelheim and Enara Bio join forces to discover cancer immunotherapies
Agreement will leverage Enara’s novel Dark Antigen discovery platform
Verona completes enrolment in pilot study of potential COVID-19 inhaler
Ensinfentrine is a first-in-class inhaled inhibitor of the enzymes phosphodiesterase 3 and 4
NICE backs GSK’s Zejula in advanced ovarian cancer
Zejula has been recommended for use in newly diagnosed ovarian cancer patients
US breakthrough designation for ligelizumab in chronic spontaneous urticaria
In a Phase IIb trial, more patients experienced complete resolution of hives with ligelizumab compared to Xolair
NHS England selects supermarket Asda to operate COVID-19 vaccination centre
Qualified pharmacists will administer the Pfizer/BioNTech vaccine at an in-store pharmacy
EU approval for ViiV’s Tivicay for children living with HIV
First dispersible tablet formulation of Tivicay now available for children in the EU
Valneva partners with Oxford Immunotec on T cell analysis for COVID-19 vaccine
Oxford Immunotec will perform T cell testing on participants receiving Valneva’s vaccine candidate
Gilead and Vir unite to find a ‘functional cure’ for hepatitis B
Companies will collaborate on a combination therapy study
Roche’s Tecentriq/Avastin combo improves survival in liver cancer
Updated analysis is from the Phase III IMbrave150 study
Diurnal seeks marketing authorisation for Chronocort in the UK
MHRA submission is based on same application filed with the EMA
Eli Lilly announces mid-stage trial success for donanemab in Alzheimer’s
Investigational antibody targets a modified form of beta amyloid called N3pG
Synairgen begins large-scale trial of inhaled COVID-19 treatment
First patient in global trial dosed in the UK