Updated study results showed an overall response rate of 98% with a longer-term follow-up at a median of 18 months
News
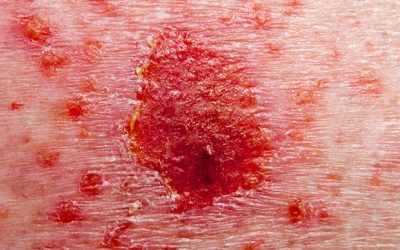
Cosentyx approved to treat plaque psoriasis in paediatric patients
IL-17A inhibitor has been cleared in the US to treat plaque psoriasis in certain patients aged six years and older
WHO authorises Sinovac’s COVID-19 vaccine for emergency use
A WHO emergency use listing (EUL) is issued when a product meets international standards for safety, efficacy and manufacturing
EC authorises Diurnal’s Efmody for CAH
First commercial launch of Efmody is expected in the third quarter of 2021
University of Bradford to host COVID-19 vaccine booster trial
Trial is being conducted as part of a £90m Department of Health and Social Care (DHSC) initiative
Sanofi halts venglustat clinical programme in ADPKD
Phase II/III trial of the drug did not meet its futility criteria of the annualised rate change in total kidney volume (TKV)
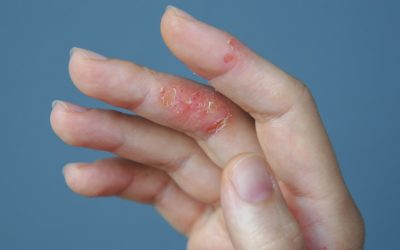
Amgen and Kyowa Kirin agree to jointly develop eczema med
KHK4083 is an anti-OX40, fully human monoclonal antibody in development for the treatment of atopic dermatitis
BREAKING NEWS: UK approves J&J’s one-dose COVID-19 vaccine
UK’s Vaccine Taskforce has secured 20 million doses of the vaccine, with initial deliveries expected ‘later this year’
NICE green lights Tremfya to treat active psoriatic arthritis
Tremfya has already been recommended for NHS use to treat eligible patient with moderate-to-severe plaque psoriasis
Novacyt disputes COVID-19 testing contract with DHSC
Clinical diagnostics company said dispute may have a material impact on its Q1 revenues
Royal College of Surgeons calls for creation of ‘surgical hubs’ to tackle backlog
Latest NHS waiting list figures show that 4.95 million people are currently waiting for elective surgery
EU clears Tagrisso for early-stage EGFR-mutated lung cancer
In Phase III, adjuvant treatment with Tagrisso reduced the risk of disease recurrence or death by 80% in the overall population
Janssen’s Erleada maintains quality of life for prostate cancer patients
New patient-reported outcomes data also shows Erleada did not worsen side effect burden
CQC unveils new strategy based on extensive external consultation
Strategy “marks a shift away from organisations and towards service improvement”
FDA approves Pfizer, Myovant’s uterine fibroids drug Myfembree
Once-daily treatment has been approved for the management of heavy menstrual bleeding